Federal Cannabis Rescheduling Is Here: What It Means for Cannabis Operators
Federal Cannabis Rescheduling Is Here: What It Means for Cannabis Operators
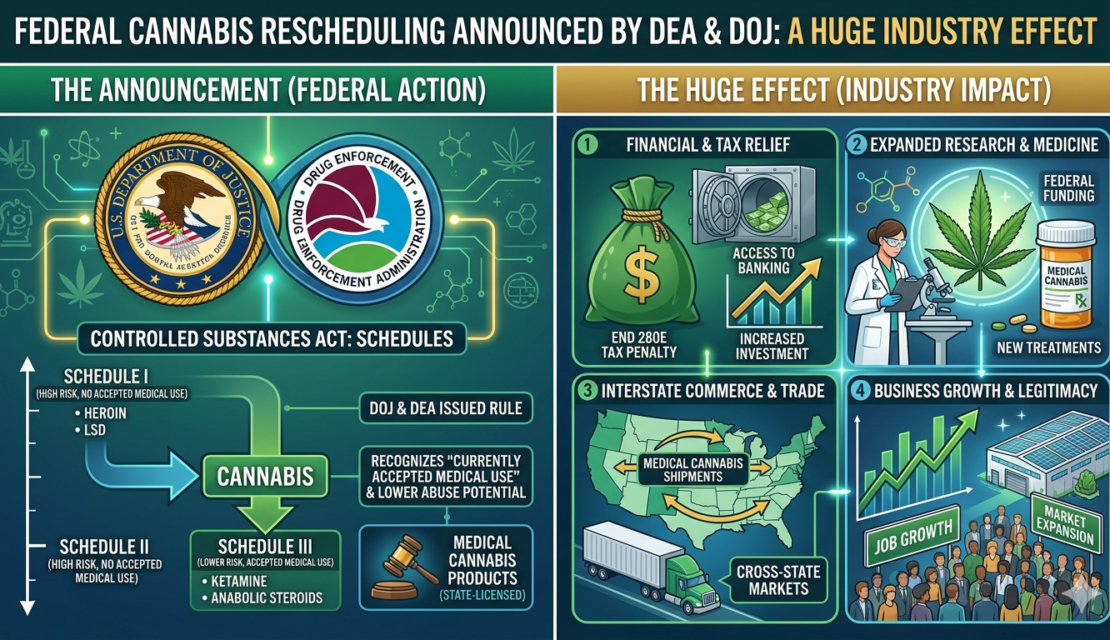
For years, cannabis operators have been running businesses under some of the most punishing tax conditions of any industry in the country. That is now starting to change. On April 23, 2026, Acting Attorney General Todd Blanche signed a final order through the Drug Enforcement Administration officially moving marijuana products regulated by state medical cannabis licenses from Schedule I to Schedule III of the Controlled Substances Act, effective April 22, 2026. This is not a proposal or a preliminary step. It is a signed, effective final order with an immediate compliance date — and operators across every license type need to understand what it requires of them right now.
In this post we break down what the order actually says and what it means for your business. We cover:
- How rescheduling affects medical operators, adult-use operators, and dual-use operators separately, because the impact and the obligations are meaningfully different depending on your license type.
- The end of Section 280E liability for medical cannabis businesses and what that means for your tax burden and banking access.
- The new federal DEA registration requirement, the fees involved, and the 60-day window operators need to act within.
- How the federal government is integrating existing state licensing systems into the new federal registry rather than replacing them — a significant and encouraging design decision.
- The new crop purchase mechanism for manufacturers.
- The new research opportunities rescheduling unlocks and why those matter for market growth,
- What this means for states that have not yet legalized, and
- How this fits into a broader federal shift toward regulated access that also includes a recent executive order on psychedelics.
Throughout all of it, we emphasize what every operator needs to hear right now:
rescheduling is not just a tax story, it is a compliance story, and the next 60 days will define
how much of the opportunity you actually capture.
How Rescheduling Is Rolling Out
The DOJ is moving in phases. Per the final order, effective April 22, 2026, marijuana products regulated by a state medical cannabis license moved immediately to Schedule III, as did any marijuana products approved by the Food and Drug Administration. The order defines a “state medical marijuana license” as a license issued by a state entity authorizing the licensee to manufacture, distribute, and/or dispense marijuana or products containing marijuana for medical purposes.
The broader question of adult-use rescheduling will go through a new expedited administrative hearing process beginning June 29. That process will determine whether cannabis sold through recreational markets also moves to Schedule III.
The order is explicit that any form of marijuana other than an FDA-approved drug product or marijuana subject to a state medical marijuana license remains a Schedule I controlled substance. Those who handle such material remain subject to all applicable Schedule I regulatory controls, administrative, civil, and criminal sanctions. The line between what is now Schedule III and what remains Schedule I is a compliance line, and crossing it inadvertently carries serious consequences.
What Rescheduling Means for Medical Cannabis Operators
For operators holding state medical cannabis licenses, April 22, 2026 marks a fundamental shift in how your business is treated under federal law. Schedule III status took effect immediately, and with it comes both meaningful relief and new federal obligations.
On the relief side, the most significant change is the end of Section 280E liability. Section 280E of the Internal Revenue Code has been the single most destructive financial burden on the legal cannabis industry since it emerged. The provision, originally written to prevent drug traffickers from claiming tax deductions, applies to any business trafficking in Schedule I or Schedule II controlled substances. Because cannabis sat at Schedule I, every state-licensed dispensary, cultivator, and processor in the country was subject to it regardless of how legally and carefully they operated.
What 280E meant in practice was that cannabis businesses could not deduct ordinary and necessary business expenses the way every other industry can. Rent, payroll, marketing, utilities, professional services — none of it was deductible. Companies were paying effective tax rates that sometimes exceeded 70 percent of their gross income. Profitable-looking businesses on paper were cash-starved in reality.
That changes now. The final order states directly that holders of state medical marijuana licenses will no longer be subject to the deduction disallowance imposed by 280E. Standard business deductions become available, and the gap between gross revenue and taxable income widens dramatically. For many operators, this is the difference between surviving the year and not.
The order also encourages the Treasury Secretary to consider providing retrospective relief from 280E liability for prior tax years in which a state licensee operated under a state medical marijuana license. That language is not a guarantee, but it opens the door to potential refunds or credits for taxes paid under 280E in previous years. Operators in mature markets have often found ways to mitigate 280E’s impact through various tax strategies, so for many the change will simplify accounting more than it transforms the bottom line. For operators in emerging markets or those who bore the full weight of 280E without workarounds, the relief will be more material. It will be interesting to see how much actual impact to cash flow and reserves this has company by company.
What rescheduling does consistently for medical operators is make the business dramatically easier to bank. Financial institutions have historically avoided cannabis businesses because of the Schedule I designation and the regulatory exposure it created. A Schedule III classification changes the risk calculus for banks meaningfully. Access to standard business checking, merchant processing, lines of credit, and eventually conventional business loans at competitive rates becomes a realistic near-term prospect. Running your business like a normal business is a competitive advantage, and rescheduling moves medical operators meaningfully closer to that reality.
On the obligations side, medical operators must now obtain federal DEA registration. This is not optional. Operating under Schedule III requires federal registration as a manufacturer, distributor, or dispenser. The DEA has designed an expedited review process that accepts your existing state medical marijuana license as the primary evidence of authorization, and entities that apply within 60 days of the order’s publication may continue operating under their state license while the application is pending. Do not let that window lapse.
What Rescheduling Means for Adult-Use Cannabis Operators
Adult-use operators are not in the same position as medical operators today, and it is important to be clear about that. As of April 22, 2026, recreational cannabis remains a Schedule I controlled substance. The final order applies only to marijuana subject to a state medical marijuana license or contained in an FDA-approved drug product. Adult-use sales, cultivation, and distribution do not fall under either category and are therefore not covered by this rescheduling action.
The June 29 administrative hearing is the mechanism through which adult-use rescheduling will be considered. If that hearing results in a favorable ruling, recreational cannabis will also move to Schedule III, extending 280E relief and DEA registration eligibility to adult-use operations. That outcome is not guaranteed, but the expedited hearing process and the current federal posture suggest that outcome is more likely than not.
Until then, adult-use operators should be doing two things. First, monitor the June 29 hearing and its aftermath closely. The decision will have immediate financial consequences for your business, and you want to be positioned to act on both the tax and banking fronts the moment rescheduling applies to your operations. Second, begin preparing now. Understand what your 280E exposure has been, what deductions you will be entitled to claim, and what your books will look like under a different tax regime. The operators who move fastest after a favorable ruling will be in position to capture the most benefit.
What Rescheduling Means for Dual-Use Operators
For operators holding licenses that cover both medical and adult-use sales, the situation is more nuanced and requires careful attention to how your operations are structured and accounted for.
Effective April 22, 2026, the medical portion of your business is now subject to Schedule III. That means 280E relief, DEA registration eligibility, and improved banking access apply to your medical revenue and the costs associated with serving medical patients. The adult-use portion of your business, however, remains Schedule I and does not benefit from any of these changes yet.
This creates an immediate and urgent compliance imperative: your medical and adult-use operations need to be clearly separated in your accounting, your record-keeping, and where possible your physical operations. The IRS and DEA will expect you to be able to distinguish, with documentation, which revenue, expenses, inventory, and activity relate to your medical license versus your adult-use license. Commingling these will create exposure on the medical side that could cost you the very relief you are entitled to, and it will create regulatory risk that compounds when adult-use rescheduling arrives in June.
Once the June 29 hearing concludes and adult-use rescheduling is approved, that Schedule III status will extend to the recreational portion of your business as well, making the full operation 280E-exempt and eligible for full DEA registration. The operators who have done the work to properly separate their operations in the interim will be able to capture that benefit immediately and cleanly. Those who have not will be scrambling to reconstruct records that should have been maintained from day one. File for DEA registration now for your medical operations. When adult-use rescheduling takes effect, you can expand your registration accordingly. Getting your federal compliance infrastructure in place on the medical side now means less friction when the recreational side follows.
Compliance Is No Longer Optional: A New Federal Layer Requires Attention
Rescheduling is not just a tax story. It is a compliance story, and that framing matters more than it is getting credit for right now. Every state-licensed medical cannabis operator is now operating under a new federal regulatory framework that did not exist 48 hours ago, and the obligations that come with it are real.
- Federal DEA registration is required.
- The crop purchase mechanism for manufacturers is required.
- DEA facility access for cultivators is required.
- Record-keeping that satisfies both state and federal standards is required.
- The federal warning label on applicable products is required.
None of these are suggestions.
The good news is that the final order was designed to minimize the compliance burden on operators who are already operating in good standing under their state licenses. State records are accepted, state certifications are sufficient for dispensing, and state security and labeling requirements satisfy most federal standards. But “minimized burden” is not the same as “no burden.” There is a new layer here, and it requires operators to understand it, adapt to it, and document their compliance with it.
The operators who treat this moment as a compliance inflection point — not just a tax break, will be in the strongest position. Those who focus only on the financial benefits and ignore the new regulatory obligations will create exposure that could cost them far more than 280E ever did. DEA registration can be suspended or revoked. State licenses can be affected by federal compliance failures. The stakes of getting this wrong have not gone down; in some ways they have gone up.
The Research Mandate: What New Medical Findings Mean for the Industry
One aspect of rescheduling that deserves more attention from operators is the research dimension. Schedule I classification has historically barred or significantly impeded formal clinical research on cannabis. With that barrier removed, the medical and scientific community will now be able to study cannabis more rigorously and at greater scale than has ever been possible.
The findings that emerge from this research will matter to the industry in ways that go beyond regulation. Peer-reviewed studies on efficacy, dosing, contraindications, and long-term outcomes will reach physicians, insurers, patients, and policymakers who have remained on the fence. Some of those findings will be cautionary and will likely deter certain users. But the broader momentum from credible, federally sanctioned research is expected to bring far more people to the table than it pushes away.
When patients who have been hesitant to try cannabis receive guidance from their physicians backed by clinical evidence rather than anecdote, the conversation changes. People who abandoned cannabis after a poor early experience, or who have avoided it due to stigma or uncertainty, will engage differently when their doctor can speak to it with the same confidence they would bring to any other Schedule III medication. That shift in credibility has the potential to expand the patient base significantly. More broadly, it opens the door to cannabis being used as medicine at an industrial scale — integrated into healthcare systems in ways that have simply not been possible until now.
What This Means for States That Have Not Yet Legalized
Federal rescheduling matters beyond the states where adult-use markets already exist. For states that still have medical-only programs or have not yet passed any form of legalization, this development changes the calculus significantly.
One of the most persistent arguments against legalization in conservative-leaning states has been the perception that cannabis remains a federally criminal enterprise. Rescheduling does not federally legalize cannabis, but it fundamentally reframes it. A Schedule III substance carries a very different political profile than something classified alongside heroin. Legislators who have faced political risk from the Schedule I designation now have considerably more cover.
The removal of 280E liability also changes the business case for potential operators and investors in unlicensed states. Capital that has been sitting on the sidelines will start to move. Texas, which just awarded 12 new dispensing organization licenses under its Compassionate Use Program expansion, is a direct example. Those licensees are entering the market at a moment when the federal tax environment has fundamentally improved, creating an unusually favorable position for operators who get up and running in the near term.
How State Licenses Are Being Integrated Into the Federal Registry
One of the more significant design decisions embedded in this final order is how the DOJ has chosen to handle the relationship between existing state licensing systems and the new federal registration framework. Rather than treating state licenses as irrelevant to federal compliance or requiring operators to rebuild their regulatory standing from scratch at the federal level, the order does something more pragmatic: it formally incorporates state licensing infrastructure into the federal framework.
The Attorney General’s order states directly that incorporating state licensing systems into the federal registration framework represents the most effective and efficient means of achieving the Controlled Substances Act’s objectives with respect to medical marijuana, while promoting medical benefits and causing the least disruption for patients and existing state systems. That framing is important. The federal government is not asserting supremacy over state cannabis programs here. It is recognizing that those programs, built over three decades across 40 states, have already developed the infrastructure needed to satisfy the public-interest objectives that underlie federal registration, including diversion prevention, product safety, record keeping, facility inspections, and oversight of licensees.
In practical terms, this means that a state-issued medical marijuana license functions as conclusive evidence of authorization under state law when applying for federal DEA registration. Operators do not need to prove to the DEA that they are authorized to operate — their state license does that work. The DEA must grant registration unless doing so would be inconsistent with the public interest or with the requirements of the Single Convention on Narcotic Drugs. For any operator maintaining a clean, compliant state license in good standing, that bar is straightforward to clear.
The order also establishes:
- That state-required records, reports, and order forms are accepted to the maximum extent permissible under federal law.
- State-authorized certifications for dispensing are sufficient for patient transactions, provided they contain the patient’s name and address, are dated and signed on the day of issuance, and identify the issuing practitioner.
- State labeling, packaging, disposal, and physical security requirements satisfy the otherwise-applicable federal standards, subject to inclusion of the federally required warning label.
In short, operators who are already meeting their state compliance obligations will not be required to build an entirely parallel compliance infrastructure for federal purposes. What you are already doing counts.
There are a few areas where new federal obligations do add a layer that state systems have not previously required.
- Manufacturers must register the specific areas in which cultivation is permitted.
- The DEA must maintain access to crop storage facilities under the purchase-and-resale mechanism described earlier.
- And federal DEA registration, once issued, automatically suspends if the underlying state license is suspended, revoked, or expires — meaning your federal standing is directly tethered to your state standing. That linkage cuts both ways: a clean state license keeps your federal registration intact, but any state compliance failure now has an immediate federal consequence as well.
What this architecture signals, beyond the operational mechanics, is a deliberate federal posture of cooperative federalism. The DOJ is building a federal framework that treats the existing state regulatory apparatus as a foundation to build on rather than an obstacle to work around. For the cannabis industry, that is meaningful. It validates the legitimacy of state licensing systems in a way that has not previously existed at the federal level, and it sets a precedent for how federal cannabis oversight is likely to evolve as additional rescheduling actions follow.
For operators, the message is this: your state compliance record is now your federal compliance foundation. Every gap in your state license history, every disciplinary action, every lapse in record-keeping becomes visible in a federal context that it previously did not. Operators who have maintained rigorous compliance with their state programs are in an excellent position. Those who have let things slide at the state level need to address that before they approach federal registration.
New Federal Registration Fees
Per the final order, operators registering with the DEA under Schedule III will be subject to the following fees:
Manufacturers: $3,699 annually. Distributors: $1,850 annually. Dispensers, including pharmacies: $888 for a registration valid for three years.
These are modest costs relative to the benefits, but operators should budget for them and prioritize filing within the 60-day window to maintain the ability to operate during the review
period.
The Bigger Picture: A Federal Shift Toward Regulated Access
Cannabis rescheduling does not stand alone as a policy development. Five days before the DOJ announcement, on April 18, President Trump signed an executive order directing federal agencies to accelerate access to psychedelic drugs as medical treatments. The order directed the FDA to prioritize review of psychedelics that have received Breakthrough Therapy designations, including psilocybin and ibogaine compounds. It also allocated $50 million through the federal Advanced Research Projects Agency for Health to partner with states advancing psychedelic research programs, and directed the DEA to establish a pathway for eligible patients to access investigational psychedelic drugs under Right to Try provisions.
Taken together, these two actions represent something notable: a federal administration moving in a consistent direction toward regulated access for substances that have long been treated as categorically off-limits. The psychedelics order is earlier in the regulatory process and does not carry the immediate commercial implications that cannabis rescheduling does. But it signals the same underlying posture: substances with demonstrated or potential medical benefit should be accessible through structured, evidence-based frameworks rather than treated as uniformly prohibited. The direction of travel is set.
What Operators Should Be Doing Right Now
Medical cannabis operators need to act immediately. Get in front of your accountant and tax attorney before the end of the quarter. File for DEA registration within 60 days of the order’s Federal Register publication. Ensure your books can distinguish medical from adult-use activity if you operate in both markets. And review your facility and record-keeping practices against the new federal requirements.
Adult-use operators should monitor the June 29 hearing closely and use the time before it to get their accounting and compliance infrastructure ready. Be ready to file for DEA registration within 60 days of June 29 hearing.
Dual-use operators have the most work to do and the most to gain. Separate your operations and accounting now. File for DEA registration on the medical side now. Prepare your compliance documentation to support both the existing medical relief and the forthcoming adult-use rescheduling.
Across all license types, the compliance dimension of this moment is as important as the financial one. New federal obligations are now in effect, and the cost of getting them wrong is high. This is exactly the kind of moment where having experienced cannabis compliance support in your corner makes a material difference.
At WeCann, we work with cannabis operators, license holders, and investors navigating these market shifts. Beyond real estate and licensing, we serve as regulatory consultants for our clients — helping them understand what new developments specifically require of their business, identify compliance gaps, and engage the right advisors on the right questions. Whether you need help understanding the DEA registration process, thinking through your medical and adult-use accounting structure, or simply want a clear read on how these changes affect the value of your assets, we are here.
Contact WeCann today. The policy environment is changing faster than at any point in the history of this industry, and your compliance posture in the next 60 days will define how much of the opportunity you actually capture.





